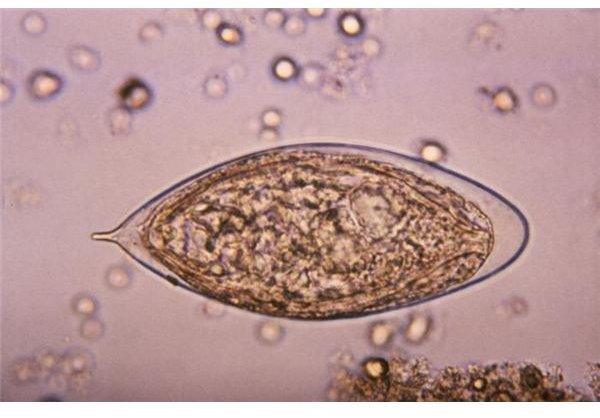
Diagnosing Schistosomiasis
Stool Examination, Biopsies, and Immunodiagnosis
Demonstrating the presence of Schistosoma eggs in the feces of a patient by direct fecal smear method is a straightforward
diagnostic technique. The problem with this method is that some species of Schistosoma like S. japonicum produce little amount of eggs that could be easily detected (using a microscope) by direct fecal smear. This problem can be solved by concentration techniques and other diagnostic methods, such as immunodiagnosis and biopsies.
One concentration technique currently available is by subjecting a stool sample into a centrifuge, a device that rotates rapidly and uses centrifugal force to separate substances of different densities. After subjecting to a centrifuge, the different compositions of the stool are separated based on their densities; the Schistosoma eggs are separated (as a distinct layer in the test tube) from other fecal particulates. The eggs would then be isolated for further laboratory examinations. With the use of this concentration technique, more than 90% of the coprologically demonstrable cases can be diagnosed.
According to Peters et al. (1980), the Kato Technique is a simple method for discovering and quantifying eggs in 20 mg to 50 mg fecal samples; although few or no eggs may be passed, specifically in chronic cases. In such cases, performing biopsies in the rectum, liver, or the bladder may be of great help as can ultrasonography (Richter 2000), however these require the services of
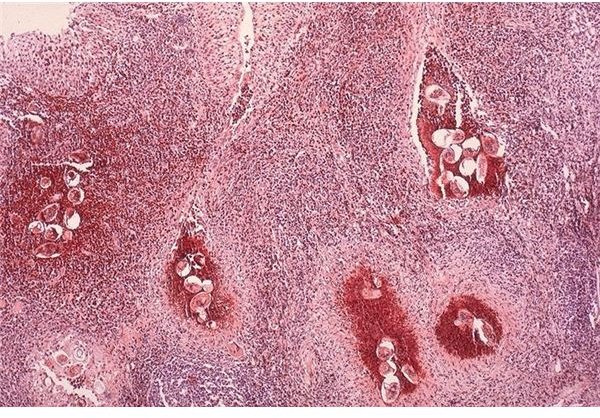
specialists and the availability of right surgical equipments. For this reason, biomedical researches are now directed at finding sensitive, accurate, and dependable immunodiagnostic methods which can be used as efficient alternative methods in diagnosing patients.
Serological Tests
Utilizing serological tests to detect antibodies in a patient’s blood sample is feasible but with several inherent problems (Strickland and Abdel-Wahab 1991). The first problem is that they (serological tests) only turn positive some time after infection; the second problem is that they only turn negative some time after treatment and; they cross-react with other helminth infections. Potential solutions to these three problems are offered by tests designed to detect schistosome antigens. The advantages of antigen detection tests are (1) they become positive as soon as antigens are present and (2) they become negative immediately after treatment. Deelder and colleagues (2000) successfully prepared monoclonal antibodies to one of the best characterized schistosome antigens. Using a modified ELISA test, they detected Schistosoma antigen in the sera of patients at levels as low as 1 ng/ml, and the concentration of antigen had high correlation with egg output; it just means that if we know the quantity of worm antigen in the serum sample we could actually estimate the number of worms per unit serum.
References
Deelder AM, Van Lieshout L, and AM Poldermann. 2000. Immunodiagnosis of schistosomiasis by determination of the circulating antigens CAA and CCA, in particular in individuals with recent or light infections. Acta Tropica 77:69-80.
Peters PA, El Alamy M, Warren KS, and AAF Mahmoud. 1980. Quick Kato smear for field quantification of Schistosoma mansoni eggs. Am. J. Trop. Med. Hyg. 29:217-219.
Richter J. 2000. Evolution of Schistosomiasis-induced pathology after therapy and interruption exposure to schistosomes: A review of ultrasonographic studies. Acta Tropica 77:111-131.
Strickland GT, and MFAbdel-Wahab.1991. Schistosomiasis. In G.T. Strickland (Ed.), Hunter’s tropical medicine, 7th ed. 81 Philadelphia: W.B. Saunders Co., pp. 781-809.
Photo Credit: https://commons.wikimedia.org/wiki/File:Schistosoma_bladder_histopathology.jpeg
https://commons.wikimedia.org/wiki/File:Schistosoma_japonicum_egg_4843_lores.jpg